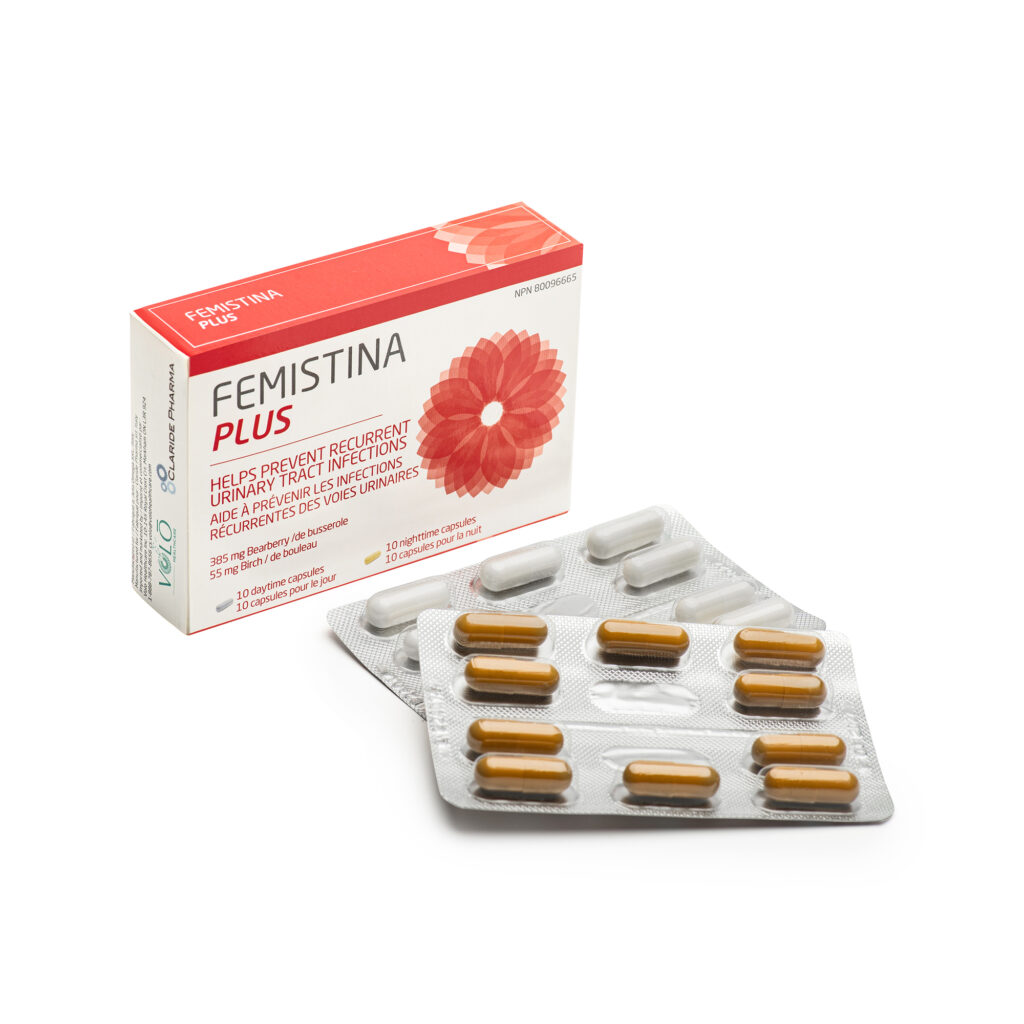
Femistina Plus provides round-the-clock defense against recurrent UTIs with its innovative triple-action mechanism. Femistina Plus is designed for use in women with a history of recurrent cystitis who are seeking a structured, non-antibiotic prophylactic strategy. It is positioned as a clinically credible adjunct or alternative to continuous low-dose antibiotic prophylaxis, aligned with global antibiotic stewardship priorities.
Mechanism of Action
Femistina Plus operates through four complementary, evidence-informed mechanisms targeting the primary pathogen responsible for recurrent UTIs — uropathogenic Escherichia coli (UPEC).
1. Anti-Adhesion via FimH Blockade (D-Mannose, 420 mg) D-mannose competitively binds to type-1 fimbriae (FimH adhesins) of UPEC, saturating bacterial adhesion sites and preventing attachment to uroepithelial cells. Bacteria unable to adhere are mechanically expelled during voiding, reducing colonization and recurrence risk.
2. Suppression of Fimbrial Expression (Berberis aristata — 98% berberine sulfate, DHE 3,000 mg) Berberine sulfate selectively inhibits the synthesis and surface assembly of Pap fimbriae on uropathogenic E. coli, reducing bacterial adhesion capacity at a molecular level. Berberine also disrupts adhesion of Streptococcus pyogenes to uroepithelial fibronectin, broadening its anti-adhesive spectrum.
3. Botanical Antimicrobial Activity (Arctostaphylos uva-ursi — 10% arbutin, DHE 1,155 mg; Betula pendula — 2.5% flavonoids, DHE 220 mg) Uva-ursi (bearberry) extract delivers arbutin, a phenolic glycoside with well-documented urinary antimicrobial activity and demonstrated prophylactic efficacy against recurrent cystitis in randomized controlled trials. Betula pendula (birch) provides complementary antimicrobial support and inhibits vaginal colonization by uropathogens.
4. cAMP-Mediated Bacterial Expulsion (Plectranthus barbatus / Forskolin — 10%, DHE 900 mg) Forskolin, a diterpene from Coleus forskohlii, elevates intracellular cyclic AMP (cAMP) levels in uroepithelial cells. Increased cAMP triggers exocytosis of intracellular bacterial reservoirs (IBCs) — the primary driver of UTI recurrence — exposing sequestered UPEC to immune clearance and antibiotic access.
Key Differentiators
- Chronobiological Dual-Capsule Architecture: Femistina Plus is the only UTI supplement in the Canadian NHP market structured as a true day/night system — the daytime capsule delivers botanical antimicrobial and anti-adhesion botanical actives; the nighttime capsule delivers D-mannose for overnight mechanical bacterial flushing. This formulation strategy optimizes pharmacological timing to address distinct phases of the infection cycle.
- Four-Target Multi-Mechanism Formula: Unlike single-ingredient products (e.g., cranberry, D-mannose alone), Femistina Plus simultaneously targets bacterial adhesion, fimbrial expression, urinary tract antimicrobial defense, and intracellular bacterial reservoir clearance — a clinically relevant multi-pathway approach.
- Clinical Evidence of Superiority Over Established Comparator: In a prospective, randomized non-inferiority study (n=320) published in the European Journal of Obstetrics & Gynecology and Reproductive Biology (Rechberger et al., 2022), Femistina demonstrated statistically significantly fewer post-surgical UTIs (6.25%) compared to Canephron N (15.63%) following midurethral sling surgery (p=0.007) — demonstrating non-inferiority and clinical utility in high-risk surgical populations.
- Antibiotic Stewardship Alignment: Femistina Plus directly addresses Canada’s and global HCP communities’ growing demand for evidence-informed, non-antibiotic prophylactic strategies for recurrent UTI — a condition that accounts for a disproportionate share of community antibiotic prescriptions and a growing proportion of antimicrobial resistance burden.
Target Prescribers / Recommenders
- Family Physicians (GPs) managing recurrent UTI in primary care
- Urologists and Urogynecologists (particularly relevant post-surgical UTI prevention)
- Obstetricians & Gynecologists
- Pharmacists (community and clinical) — primary point-of-care for NHP recommendation
- Naturopathic Doctors (NDs)
- Nurse Practitioners
Patient Profile
Primary Target: Adult women (19–65 years) with a documented history of recurrent UTIs (≥2 episodes in 12 months), particularly those experiencing cyclical antibiotic use, post-antibiotic recurrence, or antibiotic-resistant infections. Patients are typically health-conscious, motivated to reduce antibiotic dependency, and receptive to evidence-supported natural medicine.
Secondary Target: Women in peri- and postoperative urological or gynecological settings (e.g., post-midurethral sling surgery) at elevated risk of healthcare-associated UTI. Also relevant for perimenopausal and postmenopausal women in whom reduced estrogen levels increase uroepithelial vulnerability to bacterial colonization.
Clinical Characteristics: History of culture-confirmed cystitis; prior antibiotic exposure for UTI; absence of upper tract pathology, urinary stones, or stage 5 CKD; not pregnant or breastfeeding.